Deuteration-enhanced Negative Thermal Expansion and Negative Area Compressibility in a 3-Dimensional Hydrogen Bonded Network
Title: Deuteration-enhanced Negative Thermal Expansion and Negative Area Compressibility in a 3-Dimensional Hydrogen Bonded Network
Authors: Piotr Rejnhardt, Jan K. Zaręba, Andrzej Katrusiak, Marek Daszkiewicz
Journal: Chemistry of Materials
DOI: 10.1021/acs.chemmater.3c00870
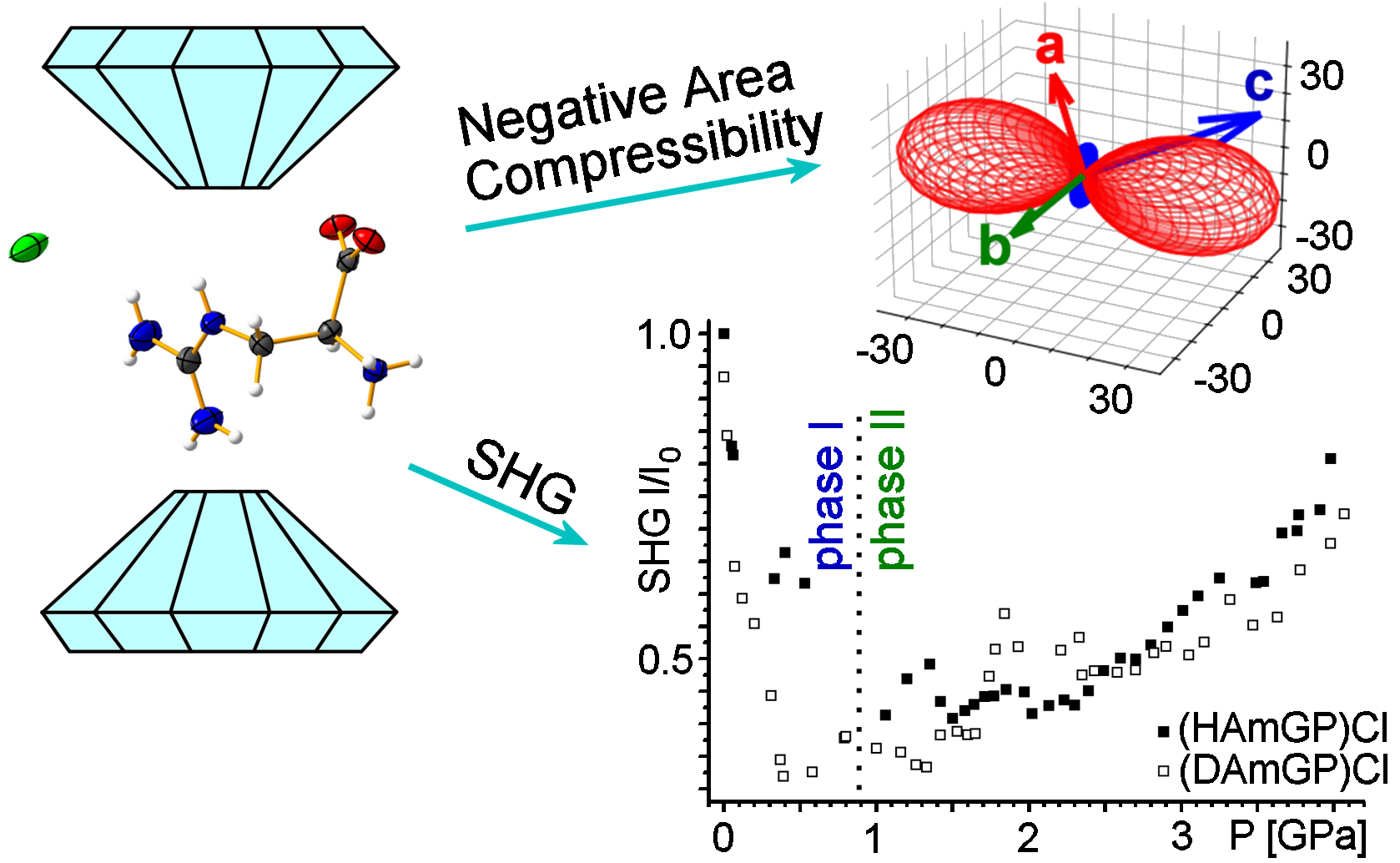
Negative area compressibility (NAC) and negative thermal expansion (NTE) are material behaviors raising high hopes for applications such as ultrasensitive manometry and thermometry. However, the group of NAC materials is primarily limited to 2D coordination compounds whose layered structure is prone to bidirectional compression when pressurized.
In this work, we reported an alternative strategy that does not employ the formation of metal-organic systems but takes advantage of the pliability of hydrogen-bonded networks for searching materials with very rare NLC and NAC behaviors. We found that strong NAC property, coupled with large NTE, has been identified for the hydrogen-bonded hydrochloride salt of L-arginine homologue (S)-2-amino-3-guanidinopropanoic acid monochloride – (HAmGP)Cl and its deuterated analogue – (DAmGP)Cl. While both structures feature isosymmetric phase transition at ca. 0.88 GPa, it is discovered that the replacement of protium with deuterium significantly boosts NAC magnitudes: phase II of (DAmGP)Cl features over two-fold higher negative area compressibility coefficient than that of nondeuterated analogue, b2,3 = –17.6(27) TPa–1 for (DAmGP)Cl vs. –7.9(38) TPa–1 for (HAmGP)Cl. Remarkably, obtained value is the second largest area compressibility coefficient known for any material. What is more, deuteration enhances also NTE property of the investigated supramolecular network, a2 = –23.4(22) MK–1 vs. –16.1(31) MK–1 for (DAmGP)Cl and (HAmGP)Cl, respectively. Thus, our results challenge the extant belief that the structure composed of interactions/bonds of extremely different nature are key to the NAC. This research opens new paths to the preparation of organic hydrogen-bonded materials with unique mechanical responsiveness to temperature and pressure stimuli.
We also observed high intensity of SHG response for both structures, (HAmGP)Cl and (DAmGP)Cl, at ambient conditions. Dramatic loss of the SHG response for phase I and its gradual restoration for phase II are observed during squeezing the samples. It appears that the three-dimensional shrink of the crystal has a negative effect on the recorded signal (phase I). Since the negative area compressibility is observed for the phase II, we suppose that molecular separation in two dimensions increases polarizability, which enhances non-linear response. All in all, the reported compounds of (S)-2-amino-3-gunidinopropanoic acid can be used as a very sensitive pressure indicators because of high values of NAC coefficients.


